Cells organize many of their most important activities using structures known as biomolecular condensates. Unlike traditional compartments in the cell, these droplet-like clusters are not enclosed by membranes. They help control how genetic instructions in DNA are converted into proteins, assist in clearing away cellular waste that could otherwise become toxic, and can even play a role in suppressing tumor growth. Because condensates behave like liquids that can fuse, flow, and quickly exchange components, scientists long believed they were simple, unstructured droplets.
However, groundbreaking research published on February 2, 2026, in the esteemed journal Nature Structural and Molecular Biology is fundamentally challenging this long-held paradigm. A dedicated team at Scripps Research has unveiled compelling evidence that certain biomolecular condensates are far from being mere amorphous blobs. Instead, these vital cellular structures are intricately constructed from complex networks of thin, thread-like protein filaments. This internal scaffolding imparts a defined architecture to these liquid-like droplets, an architectural feature that is proving to be crucial for their diverse biological functions. This discovery not only reshapes our understanding of cellular organization but also heralds the potential for entirely new therapeutic strategies to combat a spectrum of debilitating diseases, including cancers and neurodegenerative disorders.
A Paradigm Shift in Understanding Cellular Compartments
For decades, the prevailing scientific consensus viewed biomolecular condensates as dynamic, liquid-like assemblies that facilitated cellular processes through their ability to rapidly mix and exchange molecules. Their lack of a surrounding membrane, a hallmark of traditional organelles like the nucleus or mitochondria, led to the assumption that their organization was inherently fluid and unstructured. This perceived lack of distinct architecture posed a significant hurdle for therapeutic interventions.
"Ever since we realized that disruptions in condensate formation are at the heart of many diseases, it has been challenging to target them therapeutically because they appeared to lack structure — there were no specific features for a drug to latch onto," explained Keren Lasker, an associate professor at Scripps Research and the senior author of the study. "This work changes that. We can now see that some condensates have an internal architecture, and that, importantly, this structure is required for function, opening the door to targeting these membrane-less assemblies much like we target individual proteins."
This revelation marks a significant departure from the previous understanding, suggesting that the internal organization of these non-membranous compartments is not a passive consequence of molecular interactions but an actively maintained and functionally essential feature. The implications for drug development are profound, offering tangible targets for intervention where previously none were readily apparent.
Unraveling the Architecture of PopZ Condensates
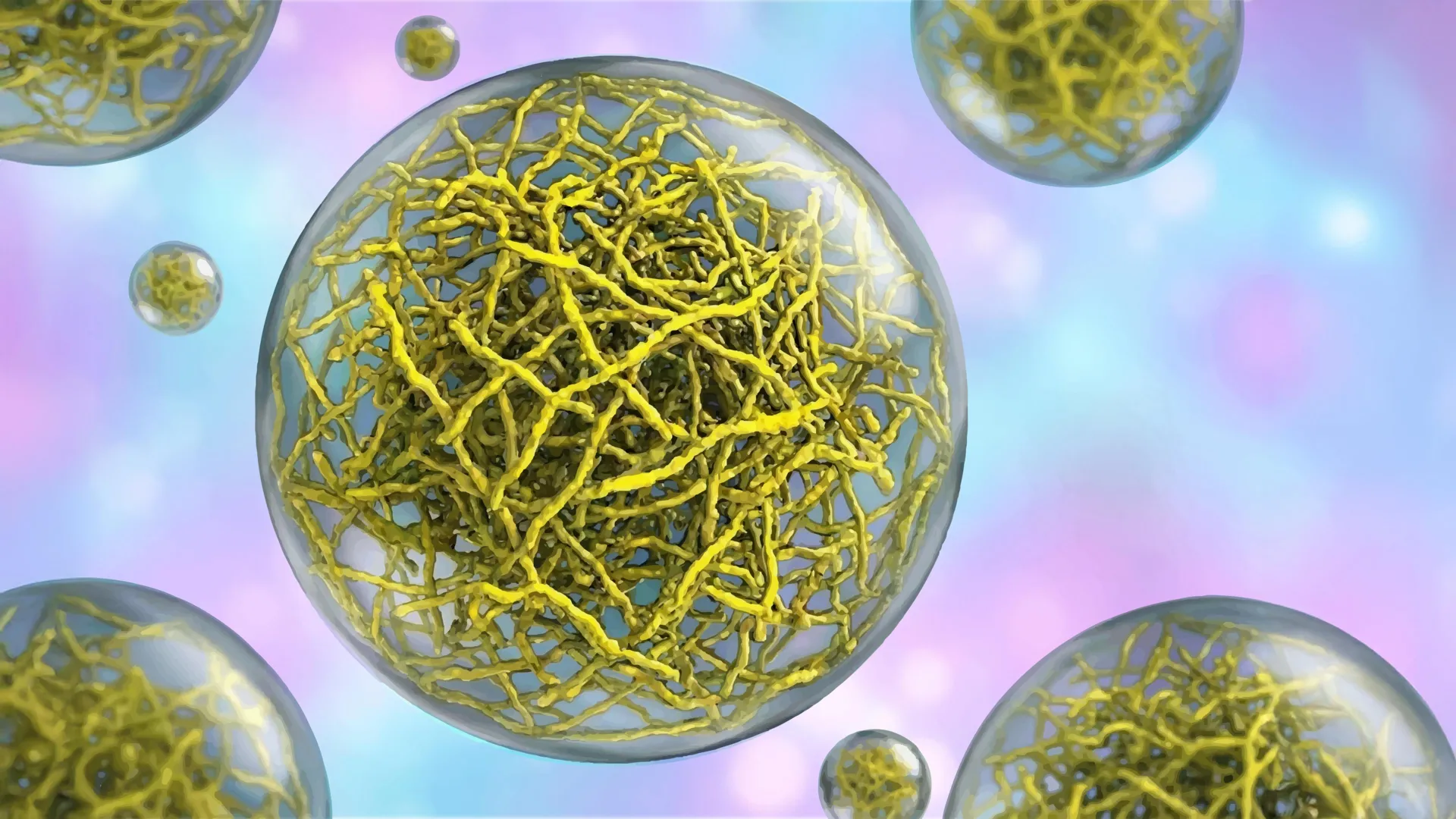
To meticulously investigate how condensates can effectively function as cellular compartments without the presence of a membrane, Lasker’s laboratory focused its attention on a well-characterized bacterial protein known as PopZ. In specific species of rod-shaped bacteria, PopZ proteins are known to localize to the cell poles – the rounded extremities of the bacterial cell. Here, they assemble into condensates that play a critical role in organizing other essential proteins required for cell division.
In close collaboration with Scripps Research professor Ashok Deniz and assistant professor Raphael Park, who co-led this pivotal study, the research team employed a powerful advanced imaging technique called cryo-electron tomography (cryo-ET). This method functions akin to a molecular-scale CT scan, providing researchers with exceptionally detailed, three-dimensional views of cellular structures at resolutions previously unattainable. The cryo-ET imaging provided unprecedented clarity, revealing that PopZ proteins do not randomly aggregate. Instead, they meticulously assemble into distinct filaments through a highly ordered, step-by-step process. These emergent filaments then coalesce to form a sophisticated scaffold, which dictates the overall physical characteristics and organizational properties of the condensate.
Dynamic Protein Conformations Inside Condensates
The investigation did not stop at the structural level. The researchers delved deeper to understand the behavior of individual PopZ molecules within and outside these condensates. Utilizing a sophisticated biophysical technique called single-molecule Förster resonance energy transfer (FRET), they were able to detect minute alterations in the distances between different parts of a protein. This was achieved by measuring the energy transfer between fluorescent tags attached to the protein. The FRET experiments yielded a remarkable finding: PopZ proteins undergo a significant conformational change – a change in their three-dimensional shape – depending on their location. The protein adopts one specific shape when it is outside a condensate and a distinctly different shape when it is incorporated within the condensate’s structure.
"Realizing that protein conformation depends on location gives us multiple ways to engineer cellular function," stated Daniel Scholl, the first author of the study and a former postdoctoral researcher in both the Lasker and Deniz labs. This observation suggests that the condensate environment actively influences the functional state of the proteins it contains, adding another layer of complexity to their role in cellular regulation. The ability of proteins to switch conformations based on their environment within the condensate opens up possibilities for fine-tuning cellular processes.
The Indispensable Role of Filament Structure
To definitively ascertain whether these internal protein filaments were merely passive structural components or actively essential for the condensate’s biological function, the research team devised a crucial experiment. They engineered a mutant version of the PopZ protein that was incapable of forming the characteristic filaments. When these altered PopZ proteins were introduced into bacteria, the resulting condensates exhibited significantly altered physical properties. They became considerably more fluid and displayed a markedly lower surface tension compared to wild-type condensates.
The functional consequences of these altered physical properties were profound. When these mutant condensates were present in living bacteria, the cells exhibited a complete failure to grow and were unable to properly segregate their DNA during cell division. This critical experiment unequivocally demonstrated that the internal filamentous structure of the condensate, and the resulting physical properties it confers, are not just decorative but absolutely vital for maintaining normal cellular function and viability. This finding moves beyond the chemical composition of condensates to highlight the importance of their physical organization.
Broader Implications for Human Health: Cancer and Neurodegenerative Diseases
While the experimental focus of this study was on a bacterial protein, the fundamental principles elucidated have broad and significant relevance to human cellular biology and disease. In human cells, filament-based condensates are known to perform two major classes of critical tasks: the meticulous clearance of damaged or misfolded proteins that could otherwise accumulate and become toxic, and the precise regulation of cell growth and proliferation.
A breakdown in the function of protein-clearing condensates is strongly implicated in the pathogenesis of a range of devastating neurodegenerative diseases, such as Amyotrophic Lateral Sclerosis (ALS). In these conditions, the cellular machinery responsible for waste removal falters, leading to the buildup of aberrant protein aggregates that damage neurons.
Conversely, when condensates that regulate cell growth malfunction, the inherent protective mechanisms that prevent uncontrolled cell division can collapse. This loss of regulation is a hallmark of cancer development, contributing to the progression of various malignancies including, but not limited to, prostate cancer, breast cancer, and endometrial cancer.
"By demonstrating that condensate architecture is both definable and functionally critical, the work raises the possibility of designing therapies that act directly on condensate structure and correct the underlying disorganization that allows disease to take hold," stated Lasker. This opens up an entirely new frontier in therapeutic development, shifting the focus from targeting individual molecules to modulating the structural integrity and functional organization of these complex cellular assemblies.
A Chronology of Discovery and Future Directions
The research leading to this groundbreaking publication represents a significant culmination of years of dedicated scientific inquiry into the fundamental nature of cellular organization. While the precise timeline of every experimental step is not publicly detailed, the progression of the research can be inferred:
- Initial Hypothesis and Exploration: The study likely began with observations suggesting that some condensates might possess more organization than previously thought, possibly driven by the known roles of certain proteins in scaffolding cellular structures. The choice of PopZ, a protein with a documented role in bacterial cell division and localization, provided a tractable model system.
- Advanced Imaging Techniques: The critical breakthrough in visualizing the internal filamentous structure would have necessitated the application and optimization of cryo-electron tomography (cryo-ET). This technique has seen rapid advancements in recent years, making it increasingly capable of resolving molecular details within complex cellular environments. The collaboration with experts in cryo-ET at Scripps Research would have been instrumental.
- Biophysical Characterization: Following the structural revelation, the focus would have shifted to understanding the dynamic behavior of the constituent proteins. Single-molecule FRET, a technique sensitive to conformational changes, would have been employed to probe the molecular mechanics of PopZ within and outside the condensate.
- Functional Validation: The experimental manipulation of PopZ’s filament-forming ability and the subsequent observation of cellular defects provided the crucial validation of the structure-function relationship. This involved genetic engineering and precise live-cell imaging to assess growth and DNA segregation.
- Broader Implications and Publication: The findings were then contextualized within the broader landscape of human disease, drawing parallels to the roles of condensates in neurodegeneration and cancer. The rigorous peer-review process for Nature Structural and Molecular Biology would have followed, leading to the publication in February 2026.
The implications of this research extend far beyond academic curiosity. The ability to target the structural integrity of condensates opens up novel therapeutic avenues. For instance, researchers might develop drugs that stabilize essential filament structures in neurodegenerative diseases or destabilize aberrant structures that contribute to cancer cell proliferation. The precise mechanisms by which these filaments form and are regulated will be a key area of future investigation. Understanding the precise interplay between protein sequence, conformational changes, and filament assembly will be crucial for designing targeted therapies.
Expert Reactions and Future Research
While direct statements from external parties are not included in the provided text, the significance of this discovery is likely to elicit considerable interest and discussion within the scientific community. Researchers in cell biology, structural biology, and drug discovery will be keen to explore the broader applicability of these findings.
Future research will undoubtedly focus on identifying similar filamentous architectures in human cell condensates and understanding their specific roles in health and disease. The development of new imaging and biochemical tools will be essential to further dissect the complex interplay between condensate structure, dynamics, and cellular function. The potential for developing novel diagnostics and therapeutics based on modulating condensate architecture represents a promising new direction in the fight against some of humanity’s most challenging diseases. This work not only deepens our fundamental understanding of life’s building blocks but also offers a tangible pathway towards more effective treatments for a wide range of human ailments.
The study, titled "The filamentous ultrastructure of the PopZ condensate is required for its cellular function," was authored by Tumara Boyd, Andrew P. Latham, Alexandra Salazar, Asma Khan, Steven Boeynaems, Alex S. Holehouse, Gabriel C. Lander, Andrej Sali, Keren Lasker, Ashok Deniz, and Raphael Park. The research received substantial support from various national and international funding bodies, including the National Institutes of Health (NINDS DP2 NS142714, NIGMS F32 GM150243, NIGMS R01 GM083960, NINDS R01 NS095892, NIGMS RO1 GM14305, NIGMS R35 GM130375, and ORIPS10 OD032467), the National Science Foundation (2235200 and DBI 2213983), the Water and Life Interface Institute, the Gordon & Betty Moore Foundation (Moore Inventor Fellowship 579361), and the Cancer Prevention and Research Institute of Texas (RR220094). This multi-faceted support underscores the broad scientific interest and recognized importance of this line of inquiry.