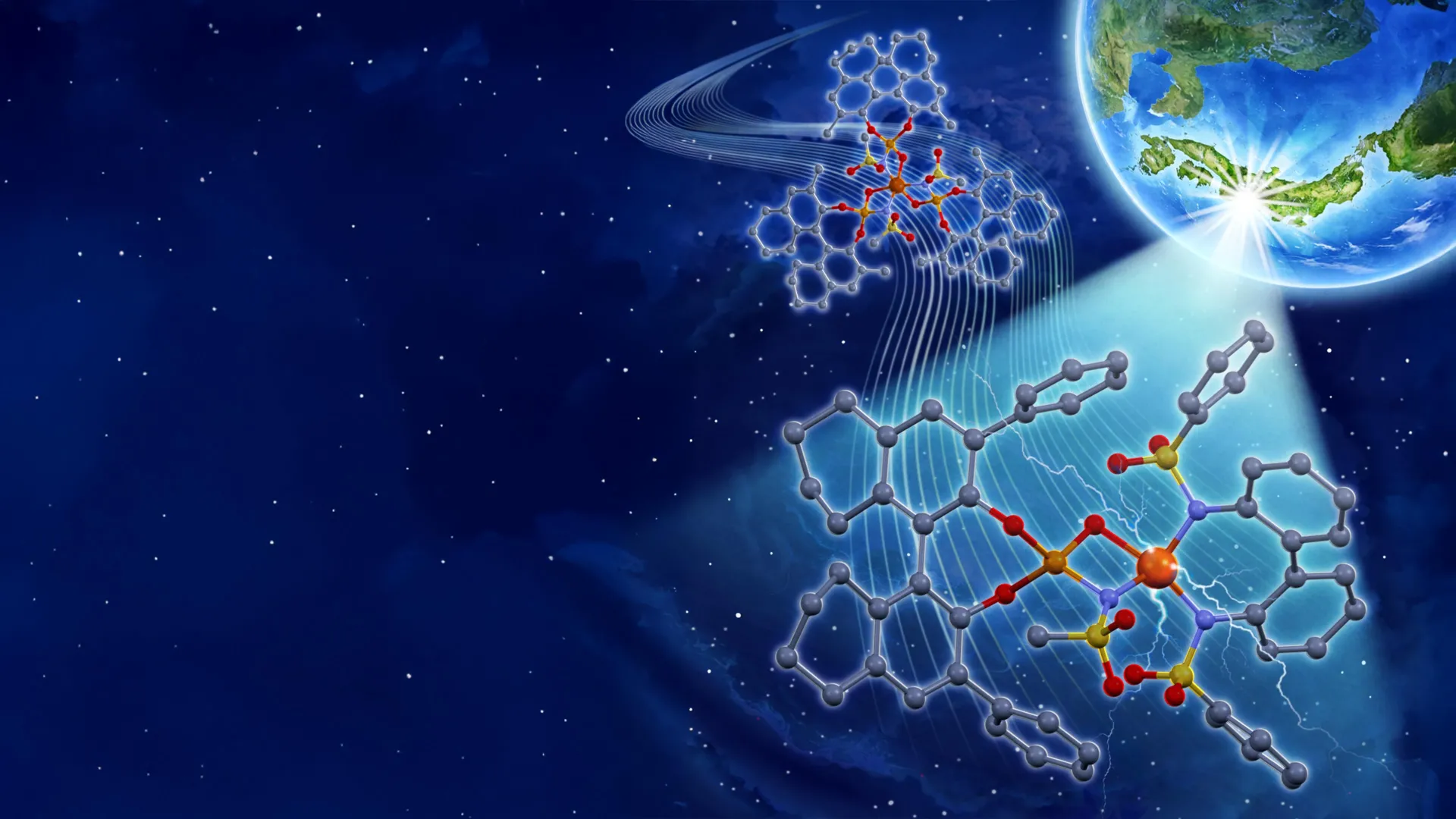
Photocatalysts are remarkable materials that harness light energy to drive chemical transformations. Within the realm of organic synthesis, metal-based photocatalysts have emerged as particularly valuable due to their inherent durability and the unparalleled ability to fine-tune their reactivity. By strategically modifying the ligands—molecules that bind to the central metal atom—chemists can precisely control the catalyst’s behavior, dictating not only the efficiency of a reaction but also the stereochemical outcome, a critical factor in the development of pharmaceuticals and other complex molecules.
Historically, many widely employed photocatalyst metals, such as ruthenium and iridium, are characterized by their scarcity and exorbitant cost. This economic and resource constraint has spurred a continuous search for more sustainable and accessible alternatives. In this ongoing pursuit, researchers at Nagoya University in Japan have achieved a significant breakthrough. Building upon their previous work introducing an iron-based substitute, the team has now unveiled a redesigned iron catalyst that dramatically reduces the reliance on costly chiral ligands, a key component in guiding the three-dimensional arrangement of synthesized chemical products. This innovative advancement promises to democratize access to complex molecular synthesis and pave the way for more sustainable chemical manufacturing processes.
A Smarter Approach to Iron Photocatalysis: Enhancing Efficiency and Sustainability
The new catalyst design, detailed in a recent publication in the prestigious Journal of the American Chemical Society, represents a paradigm shift in iron photocatalysis. In their 2023 research, the Nagoya University team had developed an iron photocatalyst that incorporated three chiral ligands per iron atom. While this approach demonstrated the potential of iron as a photocatalyst, it suffered from a significant inefficiency: only one of the three chiral ligands actively contributed to enantioselectivity—the ability to preferentially produce one mirror-image form of a molecule over another. This meant that a substantial portion of the expensive chiral ligand material was not optimally utilized, leading to higher costs and reduced overall efficiency.
The newly developed system addresses this limitation through a more strategic and elegant design. It ingeniously combines affordable, achiral bidentate ligands with chiral ligands. This synergistic combination allows for the formation of a specific iron(III) salt structure. In this optimized arrangement, the chiral ligand is strategically positioned to effectively direct the three-dimensional configuration of the final chemical product, ensuring high enantioselectivity. Concurrently, the achiral bidentate ligand plays a crucial role in enhancing the overall catalytic performance of the system, likely by stabilizing the iron center, facilitating electron transfer, or influencing the reaction environment. This dual-ligand approach not only minimizes the use of expensive chiral ligands by an impressive two-thirds compared to the previous iteration but also achieves remarkable catalytic efficiency.
Furthermore, the redesigned iron catalyst operates efficiently under the energy-saving illumination of blue light-emitting diodes (LEDs). This practical advantage significantly enhances the sustainability and economic viability of the process. Blue LEDs are widely available, consume less energy than traditional light sources, and their emission spectrum is well-suited for activating the iron photocatalyst. This shift towards more accessible and energy-efficient reaction conditions marks a significant step towards greener chemical synthesis.
Unlocking New Synthetic Pathways: The Radical Cation (4+2) Cyclization
The enhanced capabilities of this new iron photocatalyst have enabled the scientists to achieve a highly controlled radical cation (4+2) cyclization. This fundamental reaction involves the joining of two molecular components to construct a six-membered ring, a ubiquitous structural motif found in a vast array of natural products and pharmaceutical compounds. The power of this specific cyclization lies in its ability to create complex 1,2,3,5-substituted adducts, precisely the type of structural architecture that characterizes many biologically active molecules.
The development of this controlled cyclization methodology is a testament to the precise tuning of the iron catalyst’s electronic and steric properties through the judicious selection of ligands. Assistant Professor Shuhei Ohmura, one of the study’s corresponding authors, expressed strong optimism about the achievement, stating, "The new catalyst design represents the definitive form of chiral iron(III) photoredox catalysts. We believe this achievement marks a significant milestone in advancing iron-based photocatalysis." This declaration underscores the potential for this catalyst system to become a workhorse in the field, offering a robust and versatile tool for synthetic chemists.
A Landmark Achievement: First Total Asymmetric Synthesis of (+)-Heitziamide A
The impact of this improved iron photocatalyst is vividly demonstrated by its successful application in the asymmetric total synthesis of (+)-heitziamide A. Heitziamide A is a naturally occurring compound isolated from medicinal plants, recognized for its biological activity, particularly its ability to suppress respiratory bursts—a key component of the innate immune response. While previous laboratory syntheses of heitziamide A have been reported, none had achieved the total asymmetric synthesis of its naturally occurring enantiomer. This means that previous syntheses may have produced a mixture of mirror-image forms (racemic mixture) or favored the less active or inactive enantiomer.
By leveraging the stereochemical control offered by the new iron photocatalyst and the blue light-activated radical cation (4+2) cyclization, the Nagoya University team has now achieved the first-ever total asymmetric synthesis of (+)-heitziamide A. This breakthrough not only validates the synthetic power of their new catalyst but also provides a highly efficient route to this medicinally relevant natural product. The research was spearheaded by Professor Kazuaki Ishihara, Assistant Professor Shuhei Ohmura, and graduate student Hayato Akao at Nagoya University’s Graduate School of Engineering.
The significance of this achievement extends beyond the synthesis of a single compound. The researchers note that by utilizing the mirror image version of the catalyst, it would be equally feasible to produce (-)-heitziamide A. This capability allows for the selective and controlled access to both enantiomers of the molecule, a critical requirement for comprehensive pharmacological studies and the development of stereochemically pure drugs.
Implications for Pharmaceutical Chemistry and Beyond
The implications of this research for pharmaceutical chemistry and the broader chemical industry are profound. The ability to construct complex molecular architectures, including crucial pharmaceutical precursors, using abundant and inexpensive iron and readily available blue LEDs—instead of rare and costly noble metals—represents a significant leap forward in sustainable chemical manufacturing. This democratizes access to advanced synthetic methodologies, potentially lowering the cost of drug discovery and development.
Professor Kazuaki Ishihara, the study’s other corresponding author, emphasized the broader impact of their work: "Achieving the first-ever asymmetric total synthesis of (+)-heitziamide A using this catalytic reaction is a remarkable accomplishment. Several additional bioactive substances can be accessed through total synthesis, with enantioselective radical cation (4+2) cycloaddition serving as a key step. We intend to publish follow-up papers on the asymmetric total synthesis of these compounds in the near future."
This statement indicates a strategic vision to expand the application of this catalytic system to a wider range of valuable molecules. The enantioselective radical cation (4+2) cycloaddition reaction, now made more efficient and accessible, is poised to become a cornerstone methodology for accessing diverse natural products and drug candidates.
Background and Chronology of Development
The journey leading to this breakthrough began with the foundational research into iron-based photocatalysts as alternatives to precious metals. The initial efforts by the Nagoya University team, culminating in their 2023 work, established the potential of iron in this catalytic role but highlighted the need for optimization regarding chiral ligand usage.
- Pre-2023: Extensive research in photocatalysis primarily focused on noble metals like ruthenium and iridium due to their established catalytic activity and tunable properties. However, their scarcity and high cost posed significant limitations for large-scale applications.
- 2023: Researchers at Nagoya University reported the development of an iron-based photocatalyst. This was a significant step towards a more sustainable alternative, but the catalyst relied on a relatively high loading of costly chiral ligands for enantioselectivity.
- Recent Study (Published in Journal of the American Chemical Society): The team unveiled a redesigned iron catalyst. This new generation of catalyst significantly reduced chiral ligand usage by two-thirds compared to the previous version. It also demonstrated efficient operation under blue LED light, improving practicality and sustainability.
- Key Application: The redesigned catalyst was successfully employed in the first total asymmetric synthesis of (+)-heitziamide A, a naturally occurring bioactive compound.
Supporting Data and Analysis
The core of the innovation lies in the strategic combination of achiral and chiral ligands. While specific quantities and types of ligands were not detailed in the provided text, the reported reduction in chiral ligand use by two-thirds directly translates to significant cost savings and a more environmentally friendly process. The use of blue LEDs, with typical wavelengths around 450-495 nm, provides sufficient energy to excite the iron(III) complex, initiating the catalytic cycle.
The efficiency of the radical cation (4+2) cyclization is critical. This reaction class is known for its ability to form complex cyclic structures in a single step, minimizing synthetic routes and potentially improving overall yield. The success in achieving a "highly controlled" cyclization implies high diastereoselectivity and enantioselectivity, meaning the reaction preferentially forms the desired stereoisomer with minimal formation of unwanted byproducts.
The implications for pharmaceutical development can be quantified by considering the complexity of molecules that can now be synthesized more efficiently. Molecules like heitziamide A, with their intricate stereochemistry, often require lengthy and expensive synthetic pathways. A more direct and enantioselective route can drastically reduce development timelines and manufacturing costs. For instance, if a drug candidate requires a specific chiral center, an efficient asymmetric synthesis can avoid costly chiral separation steps that might be necessary if a racemic mixture were produced.
Broader Impact and Future Directions
The impact of this research resonates across several scientific disciplines. In organic chemistry, it provides a powerful new tool for constructing complex molecules with high stereochemical precision. In medicinal chemistry, it opens doors to the efficient synthesis of natural products and drug candidates that were previously difficult or economically unfeasible to produce. In materials science, the principles of designing efficient and sustainable photocatalysts can be applied to other areas, such as solar energy conversion and environmental remediation.
The Nagoya University team’s assertion that this represents the "definitive form of chiral iron(III) photoredox catalysts" suggests a high degree of confidence in the robustness and versatility of their design. The mention of future publications focusing on the asymmetric total synthesis of other bioactive substances indicates a proactive research agenda to fully exploit the potential of this catalytic system. This could include compounds with applications in oncology, infectious diseases, or neurodegenerative disorders, where stereochemistry often plays a critical role in efficacy and safety.
The development of iron-based photocatalysts, driven by the need for sustainability and cost-effectiveness, is a burgeoning field. This work by Ishihara, Ohmura, and Akao stands as a beacon, demonstrating that high-level synthetic chemistry can be achieved using earth-abundant elements and energy-efficient methodologies. The transition from expensive noble metals to abundant iron is not merely an economic advantage; it is a crucial step towards a more sustainable and responsible chemical industry. The successful demonstration of the first total asymmetric synthesis of (+)-heitziamide A serves as a compelling proof-of-concept, signaling a new era in photocatalytic organic synthesis.