Scientists have unveiled a remarkable evolutionary strategy employed by viruses that infect bacteria, revealing how these microscopic invaders effectively dismantle MurJ, a protein indispensable for constructing the bacterial cell wall. Astonishingly, diverse viruses have independently evolved distinct protein mechanisms that all converge on blocking MurJ in the exact same manner. This discovery not only illuminates a critical vulnerability in bacterial defense systems but also highlights MurJ as a highly promising new target for the development of urgently needed antibiotics. The groundbreaking findings were published on February 26 in the prestigious scientific journal Nature.
The research, spearheaded by Yancheng Evelyn Li, a graduate student within the laboratory of Professor Bil Clemons at the California Institute of Technology (Caltech), offers a significant advancement in the ongoing global battle against antimicrobial resistance. Professor Clemons, who holds the Arthur and Marian Hanisch Memorial Professorship of Biochemistry, served as the corresponding author for the study.
The Escalating Crisis of Antibiotic Resistance
The urgent need for novel therapeutic strategies against bacterial infections cannot be overstated. Bacteria possess an extraordinary capacity for rapid evolution, a trait that fuels a deepening public health crisis worldwide. As Professor Clemons articulated, "Evolution is powerful, and in bacteria, resistance to antibiotics develops quickly. This means that we now deal with bacteria that are resistant to all the medicines that we have." This growing phenomenon has dire consequences, with Professor Clemons adding, "In the USA alone, tens of thousands of people die every year from antibiotic-resistant bacterial infections, and that number is rising rapidly. We need new antibiotics to combat this."
The diminishing efficacy of existing antimicrobial drugs has compelled researchers to meticulously search for entirely new bacterial vulnerabilities. This pursuit involves delving into fundamental biological processes that are critical for bacterial survival and proliferation.
Targeting the Achilles’ Heel: The Bacterial Cell Wall
A long-standing and fruitful area of investigation has been the intricate pathway bacteria utilize to synthesize peptidoglycan, the rigid yet flexible polymer that forms the structural backbone of their cell walls. This process, known as peptidoglycan biosynthesis, presents an exceptionally attractive target for antimicrobial intervention. The primary reason for this allure lies in its specificity: peptidoglycan is a hallmark component of bacterial cell walls and is entirely absent in human cells. "Peptidoglycan is a unique feature of bacteria, and that makes it an attractive antibiotic target," Professor Clemons emphasized.
Historically, several classes of antibiotics have successfully disrupted this vital pathway. Penicillin, the groundbreaking discovery by Alexander Fleming in the mid-20th century, operates by inhibiting a late-stage enzyme involved in peptidoglycan cross-linking. Similar drugs, such as amoxicillin, function through analogous mechanisms, underscoring the therapeutic potential of targeting this pathway.
The Crucial Trio: MraY, MurG, and MurJ
Within the peptidoglycan biosynthesis pathway, a critical set of proteins facilitates the translocation of peptidoglycan building blocks across the bacterial inner membrane. Among these, MraY, MurG, and MurJ play indispensable roles, ensuring that the essential components are delivered to the exterior of the inner membrane where cell wall assembly occurs. The failure of any one of these proteins would halt peptidoglycan production, inevitably leading to the death of the bacterium. Consequently, these proteins have been identified as highly promising drug targets.
While significant progress has been made in understanding the general functions of these proteins, Professor Clemons noted that "important mechanistic details remain unclear." This knowledge gap has historically limited the development of drugs directly targeting these specific molecules.
Currently, no approved drugs directly inhibit the function of MraY, MurG, or MurJ. Nevertheless, Professor Clemons expressed optimism about future possibilities. "We do know that we can find small molecules, either derived from nature or synthesized in chemical libraries, that will inhibit these proteins. Excitingly, recent discoveries have shown that bacteriophages have figured out how to target this pathway." This insight from natural systems provided a crucial impetus for the current research.
Bacteriophages: Nature’s Bacterial Attackers
Bacteriophages, commonly referred to as phages, are viruses that specifically infect bacteria. To complete their life cycle, phages must successfully enter a bacterial cell, replicate their genetic material, and then egress to infect new host cells. The critical step of exiting the bacterial cell necessitates breaching the formidable peptidoglycan layer. Professor Clemons explained, "Getting back out means that they have to get past the peptidoglycan layer. Because it acts like chainmail, the phages get stuck if they can’t break through it." This evolutionary pressure has driven the development of sophisticated mechanisms within phages to overcome bacterial defenses.
The Clemons lab has a particular focus on studying small phages that harbor single-stranded DNA or RNA genomes. These viruses, characterized by their compact genetic material, rely on elegant and efficient strategies to induce bacterial lysis. In a notable prior study published in Science in 2023, the team reported on the intricate mechanisms of phi X174, a phage with a long and storied research history at Caltech.
Viral Proteins That Precisely Disable MurJ
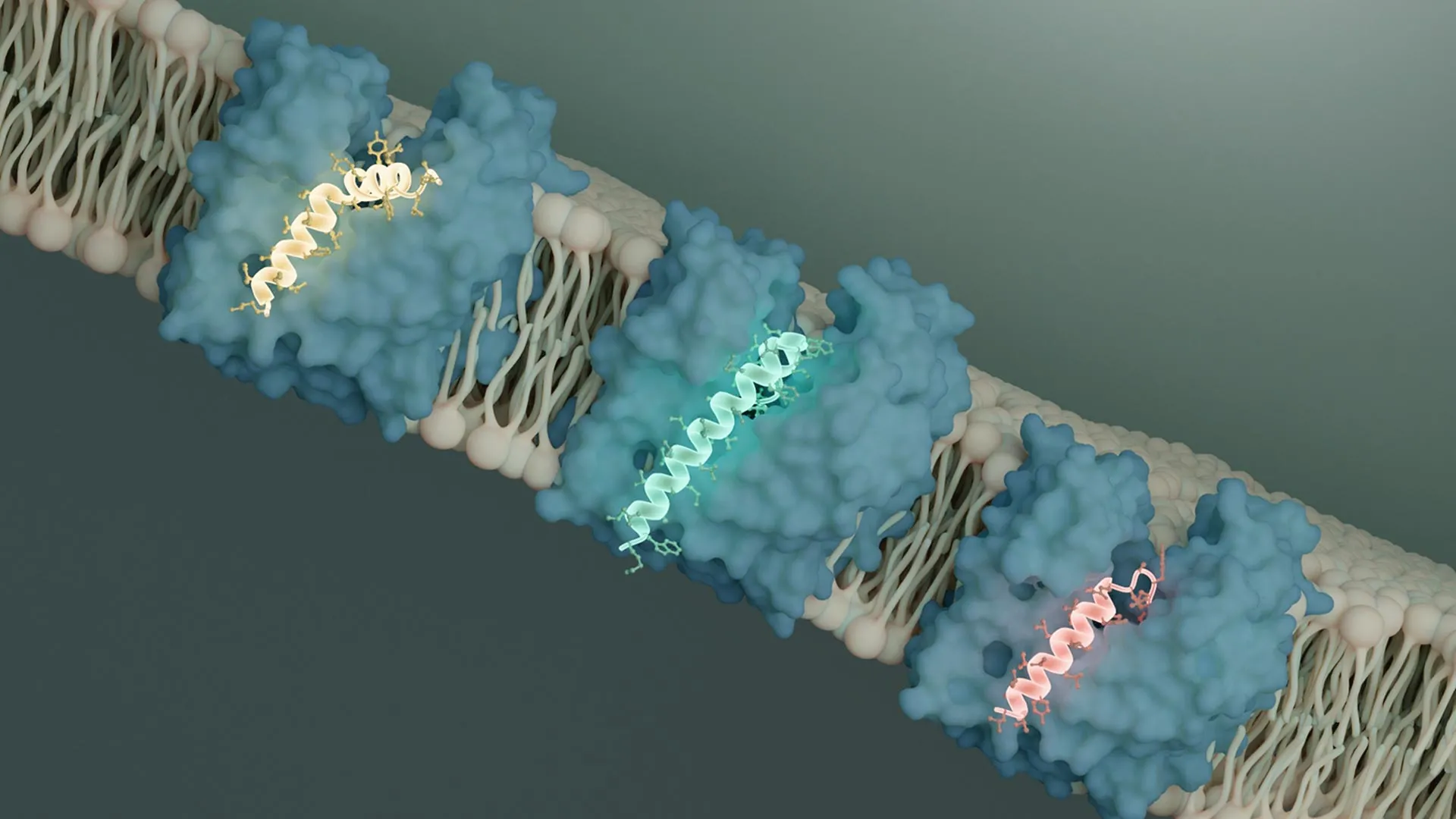
The research detailed in Nature zeroes in on a class of specialized protein antibiotics produced by small phages, known as single-gene lysis proteins, or Sgls (pronounced "sigils"). These Sgls are crucial for the phage’s ability to lyse the bacterial host and escape. Li and Clemons specifically investigated Sgls that target MurJ, one of the key proteins involved in cell wall construction.
MurJ functions as a flippase, a type of transporter protein that facilitates the movement of specific molecules across a membrane. In this context, MurJ transports peptidoglycan precursors from the cytoplasm of the bacterial cell across the inner membrane, making them available for incorporation into the growing cell wall. Previous research conducted by collaborators had already established that two evolutionarily distinct Sgls, named SglM and SglPP7, achieve bacterial killing by inhibiting MurJ.
To elucidate the precise molecular mechanism by which these viral proteins interfere with MurJ, Li employed state-of-the-art cryo-electron microscopy (cryo-EM) at Caltech’s Beckman Institute Biological and Cryogenic Transmission Electron Microscopy (Cryo-EM) Resource Center. Flippases like MurJ operate by undergoing conformational changes, effectively "flipping" to expose the transported molecule to either side of the membrane without creating a permanent pore. When MurJ binds its peptidoglycan precursor cargo within the cell, it undergoes a structural alteration to release the molecule on the external side of the membrane.
Through detailed cryo-EM structural analysis, Li made a pivotal discovery: both SglM and SglPP7 bind to a specific groove on the MurJ protein. This binding event physically obstructs the necessary structural rearrangement, or conformational shift, that MurJ must undergo to transport its cargo.
"It is clear that both of these Sgls bind to MurJ in an outward-facing conformation, locking it into this position," Li stated. This observation is particularly encouraging for therapeutic development because the outward-facing conformation of MurJ is accessible from the extracellular environment. This accessibility could make it a more straightforward target for future drug candidates compared to conformations that face inward, into the cell.
Convergent Evolution: A Testament to MurJ’s Vulnerability
Professor Clemons highlighted the profound significance of this finding, noting the surprising nature of the evolutionary convergence. "These peptides, which have no evolutionary links to each other, have both figured out how to target MurJ in a very similar way. These are two examples of convergent evolution, in which different evolutionary paths arrive at the same solution. We were surprised!" Convergent evolution, a phenomenon where unrelated organisms independently evolve similar traits or solutions to similar environmental challenges, strongly suggests that MurJ is a particularly critical and exploitable target for bacterial demise.
Given the rapid evolutionary pace of viruses, the research team posits that a vast number of other phages likely harbor similar Sgl proteins. The relative ease with which phages can be isolated and their genomes sequenced presents a rich opportunity for uncovering additional biological insights and identifying novel antibiotic targets.
In the Nature study, the researchers expanded their investigation by analyzing another phage genome with the assistance of a collaborator. This analysis led to the identification of a novel Sgl, designated SglCJ3, derived from a predicted phage genome named Changjiang3. Employing cryo-electron microscopy once again, Li meticulously determined the three-dimensional structure of SglCJ3 in complex with MurJ. The results were consistent with previous findings: SglCJ3 also functions by locking MurJ into the same outward-facing conformation, thereby inhibiting its transport activity.
"This is a third genome that evolved a distinct peptide to inhibit the same target in a similar way," Professor Clemons remarked. "It is the first strong evidence that evolution identifies MurJ as a great target for killing bacteria, which means we should follow evolution’s lead and develop therapeutics that target MurJ. This demonstrates the power of basic biology to help us solve problems in medicine. Our path is set on leveraging Sgl discovery, and we hope to continue to be supported to turn these concepts into realities."
Broader Implications and Future Directions
The implications of this research extend far beyond the immediate discovery of novel Sgl proteins. The identification of MurJ as a consistently targeted vulnerability across diverse viral lineages strongly validates it as a prime candidate for antibiotic development. The consistent evolutionary pressure exerted by different phages on MurJ suggests that targeting this protein could lead to drugs with broad-spectrum efficacy against a wide range of bacterial pathogens.
The study also underscores the immense value of exploring the natural world for solutions to pressing medical challenges. Bacteriophages, with their billions of years of co-evolution with bacteria, represent a vast and largely untapped reservoir of potential antimicrobial agents and strategies. Future research endeavors are likely to focus on:
- Expanding the Sgl Library: Systematically screening phage genomes for additional Sgls targeting MurJ and other essential bacterial proteins.
- Structure-Activity Relationship Studies: Detailed analysis of the interactions between Sgls and MurJ to optimize the design of small molecule inhibitors.
- Developing Phage-Derived Therapeutics: Investigating the direct therapeutic application of phages or their lysis proteins as a viable alternative to traditional antibiotics.
- Understanding Resistance Mechanisms: Studying how bacteria might evolve resistance to MurJ-targeting agents, a crucial step in developing robust and long-lasting treatments.
The collaborative nature of this research, involving institutions like Texas A&M University, highlights the importance of interdisciplinary approaches in tackling complex scientific problems. The funding support from organizations such as the Chan Zuckerberg Initiative and the National Institutes of Health is critical for sustaining this vital area of research.
The findings published in Nature represent a significant leap forward in our understanding of bacterial cell wall biology and the intricate arms race between bacteria and the viruses that infect them. By deciphering these evolutionary strategies, scientists are not only gaining fundamental biological knowledge but also paving the way for a new generation of antimicrobial therapies that could prove crucial in combating the ever-growing threat of antibiotic-resistant infections.
Authorship and Funding Acknowledgements
The research paper, titled "Convergent MurJ flippase inhibition by phage lysis proteins," lists Yancheng Evelyn Li and Bil Clemons as key contributors. Additional authors include Grace F. Baron, a graduate student at Caltech, and Francesca S. Antillon, Karthik Chamakura, and Ry Young from Texas A&M University. The study received generous financial support from the Chan Zuckerberg Initiative, the National Institutes of Health, the G. Harold and Leila Y. Mathers Foundation, and the Center for Phage Technology at Texas A&M, which is jointly sponsored by Texas A&M AgriLife. This collective effort and support underscore the global commitment to finding solutions for the critical challenge of antimicrobial resistance.