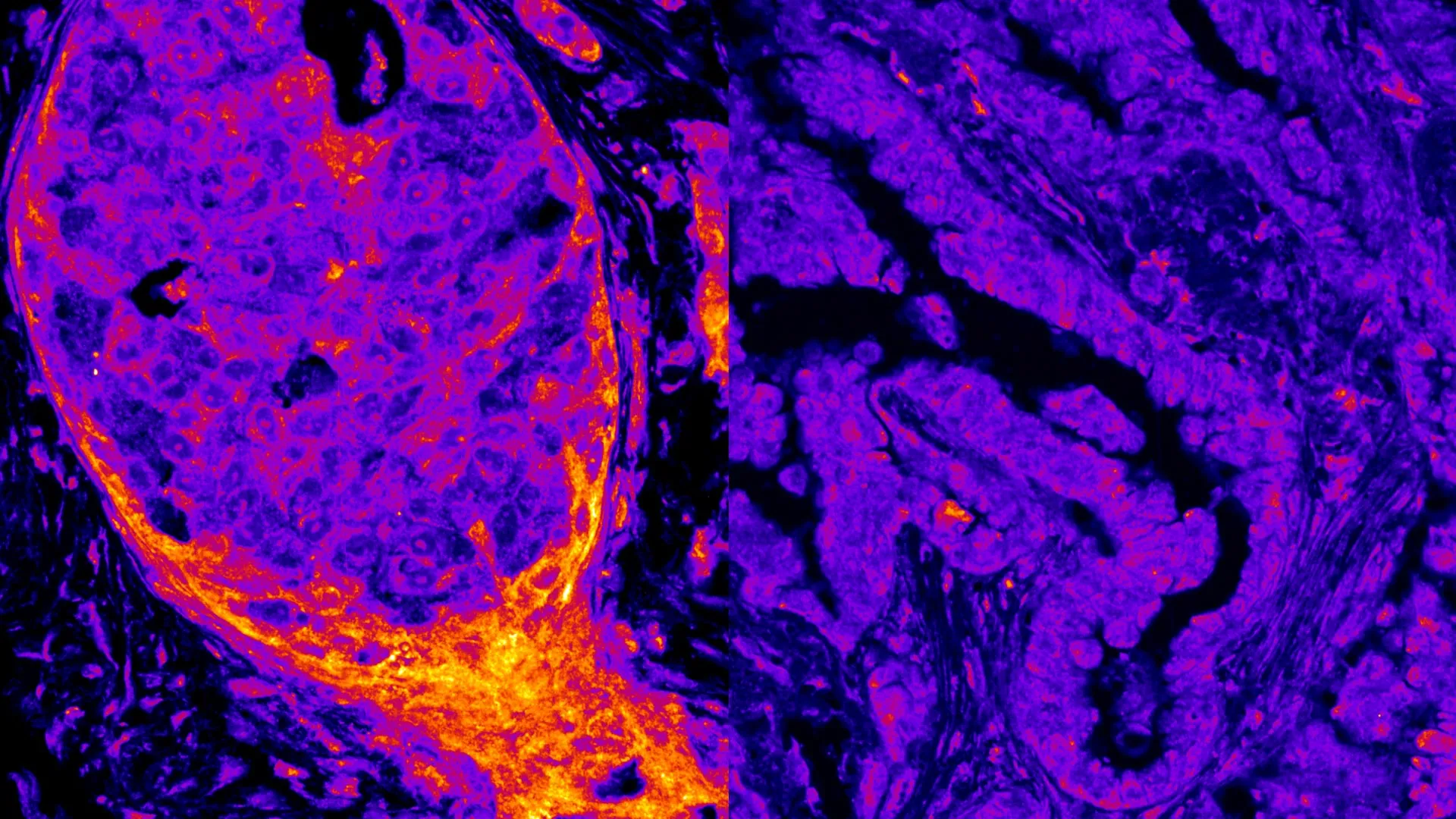
A groundbreaking study published in the prestigious journal Nature Communications has unveiled a stunning revelation: over 200 metabolic enzymes, crucial for energy production and cellular function, are not confined to their traditional cellular locations but reside directly on human DNA within the cell nucleus. This discovery challenges decades of established biological paradigms, suggesting a far more intricate interplay between metabolism and genome regulation than previously imagined, with profound implications for understanding cellular processes, disease development, and potential therapeutic interventions.
A Paradigm Shift in Cellular Localization
For years, the scientific community has operated under the assumption that the nucleus, the cell’s command center housing the genome, and the cytoplasm and mitochondria, the powerhouses of energy production, were largely separate domains with distinct functional roles. Metabolic enzymes were primarily understood to operate within the cytoplasm and mitochondria, orchestrating the complex pathways that convert nutrients into usable energy for the cell. However, the new research, spearheaded by scientists at the Centre for Genomic Regulation (CRG), has dismantled this long-held notion.
The study employed an innovative technique to isolate proteins physically bound to chromatin, the intricate complex of DNA and proteins that packages the genetic material within the nucleus. This meticulous approach allowed researchers to identify a significant number of metabolic enzymes, including those typically associated with energy generation through oxidative phosphorylation, as regular inhabitants of the nuclear environment, intimately associated with DNA. The sheer scale of this finding—approximately 7% of all proteins identified as being attached to chromatin were metabolic enzymes—points towards the existence of a sophisticated, internal metabolic network within the nucleus, a phenomenon the researchers have dubbed ‘mini metabolism.’
Unveiling the Nuclear Metabolic Fingerprint
Perhaps one of the most compelling aspects of this research is the identification of distinct "nuclear metabolic fingerprints." The study demonstrated that the composition and arrangement of these nuclear metabolic enzymes vary significantly across different cell types, tissues, and even in cancerous cells. This unique signature, observed in the patterns of enzyme-DNA interactions, represents the first concrete evidence that human cells possess such distinctive internal nuclear profiles.
"Different cell types, tissues, and cancers each display their own distinctive arrangement of metabolic enzymes within the nucleus," explained Dr. Sara Sdelci, the corresponding author of the study and a researcher at the CRG. "These enzymes interact with DNA in patterns that researchers describe as a ‘nuclear metabolic fingerprint,’ marking the first evidence that human cells may carry such unique nuclear signatures."
The implications of these unique fingerprints are far-reaching. They suggest that the nuclear metabolic landscape could play a critical role in determining a cell’s identity, function, and its susceptibility to disease. For instance, the study observed a notable difference in the prevalence of oxidative phosphorylation enzymes between breast and lung cancer cells, with breast cancer cells exhibiting a higher concentration. This variation was further validated when analyzing tumor samples directly from patients, confirming that nuclear metabolism is not only tissue-specific but also disease-specific.
The Enigmatic Roles of Nuclear Metabolites
While the discovery of these nuclear enzymes is monumental, their precise functions within the nucleus remain a subject of intense investigation. Scientists propose several possibilities: these enzymes could be directly driving biochemical reactions within the nucleus, influencing gene expression by modulating the accessibility of DNA, or even contributing to the structural integrity of the chromatin.
"Scientists still need to determine the precise role these enzymes play in the nucleus," Dr. Sdelci elaborated. "They could be driving chemical reactions, influencing whether genes are switched on or off, or contributing to structural support. Even so, the findings already provide new insights into how tumors develop, adapt, and sometimes resist therapy."
One promising avenue of research focuses on the enzymes involved in DNA repair. The study found that a group of enzymes responsible for synthesizing molecules essential for DNA replication and repair congregate at sites of DNA damage. This localized accumulation strongly suggests that these nuclear metabolic enzymes actively participate in repairing the genome, highlighting a direct link between metabolic processes and the maintenance of genetic integrity.
A New Perspective on Cancer Biology
The implications for cancer research and treatment are particularly significant. Cancer cells are notorious for their metabolic dysregulation, often exhibiting altered energy production and nutrient utilization to fuel their rapid proliferation. The presence of metabolic enzymes directly on DNA within the nucleus opens up a new frontier in understanding how cancer cells exploit these nuclear metabolic capabilities to survive and thrive.
"Many of these enzymes synthesize essential building blocks of life, and their nuclear localization is associated with DNA repair," stated Dr. Sdelci. "Their presence in the nucleus may therefore directly shape how cancer cells respond to genotoxic stress, a hallmark of many chemotherapeutic treatments. It’s an entirely new world to explore."
The findings could help explain why tumors with similar genetic mutations can respond differently to therapies. If metabolic processes and genome regulation are intricately linked within the nucleus, targeting one could inadvertently affect the other, leading to varied treatment outcomes. This interconnectedness suggests that therapies could be designed to simultaneously disrupt metabolic pathways and DNA repair mechanisms within cancer cells, potentially leading to more effective cancer treatments.
Unraveling the Mysteries of Nuclear Entry
A perplexing question arising from this research is how these often large metabolic enzymes manage to traverse the nuclear envelope, the protective barrier surrounding the nucleus. The nuclear pores, the established gateways for molecular transport, are believed to have size limitations that would typically prevent many of these enzymes from entering.
"Another unanswered question involves how these enzymes reach the nucleus in the first place," noted Dr. Savvas Kourtis, the first author of the study. "The nucleus is separated from the cytoplasm by a barrier that normally limits which molecules can pass through nuclear pores. Many of the enzymes discovered on DNA are significantly larger than the size these pores are believed to allow. Despite this, the bulky proteins still manage to enter the nucleus."
This observation strongly suggests the existence of an unknown cellular mechanism responsible for the translocation of these substantial proteins into the nucleus. Elucidating this process could reveal novel therapeutic targets for modulating nuclear metabolic activity in diseased cells, offering a new strategy for drug development.
A Timeline of Discovery and Future Directions
The research leading to this pivotal discovery likely involved years of meticulous experimentation and analysis. The initial conceptualization of investigating proteins bound to chromatin would have been followed by rigorous methodology development. The collection of diverse cell lines and tissue samples would have been a substantial undertaking, requiring careful preservation and processing. The subsequent proteomic analysis to identify and quantify the bound enzymes would have demanded advanced bioinformatics tools and expertise.
The identification of specific enzyme families, such as those involved in oxidative phosphorylation, would have been a significant milestone. The subsequent experiments demonstrating enzyme recruitment to DNA damage sites would have provided crucial functional insights. The comparison between cell types and cancer lines would have revealed the unique "nuclear metabolic fingerprints." Finally, the investigation into the differential function of enzymes based on their cellular location, like IMPDH2, would have underscored the importance of compartmentalization.
Looking ahead, the researchers emphasize that this study represents the beginning of a new era in understanding nuclear metabolism. The immediate priorities include confirming the activity of all identified nuclear enzymes and delineating their specific roles.
"Each enzyme may have its own, unique nuclear function, so this must be addressed one by one," stated Dr. Kourtis.
The long-term implications include the potential for developing novel diagnostic biomarkers for cancer based on these unique nuclear metabolic fingerprints. Furthermore, identifying new vulnerabilities within these nuclear metabolic networks could lead to the design of highly targeted anti-cancer drugs.
The scientific community is abuzz with the implications of this research. Dr. Maria Rodriguez, a leading oncologist not involved in the study, commented, "This is a truly transformative finding. If metabolism and genome regulation are so intimately connected within the nucleus, it changes how we must think about cancer development and treatment. We may need to rethink our drug development strategies and consider a more integrated approach."
This discovery, born from a meticulous investigation into the fundamental components of our cells, promises to reshape our understanding of life itself and offers a beacon of hope for more effective strategies against some of humanity’s most challenging diseases. The intricate dance between metabolism and the genome, now revealed to be occurring within the very heart of our cells, is a testament to the enduring complexity and wonder of biological systems.