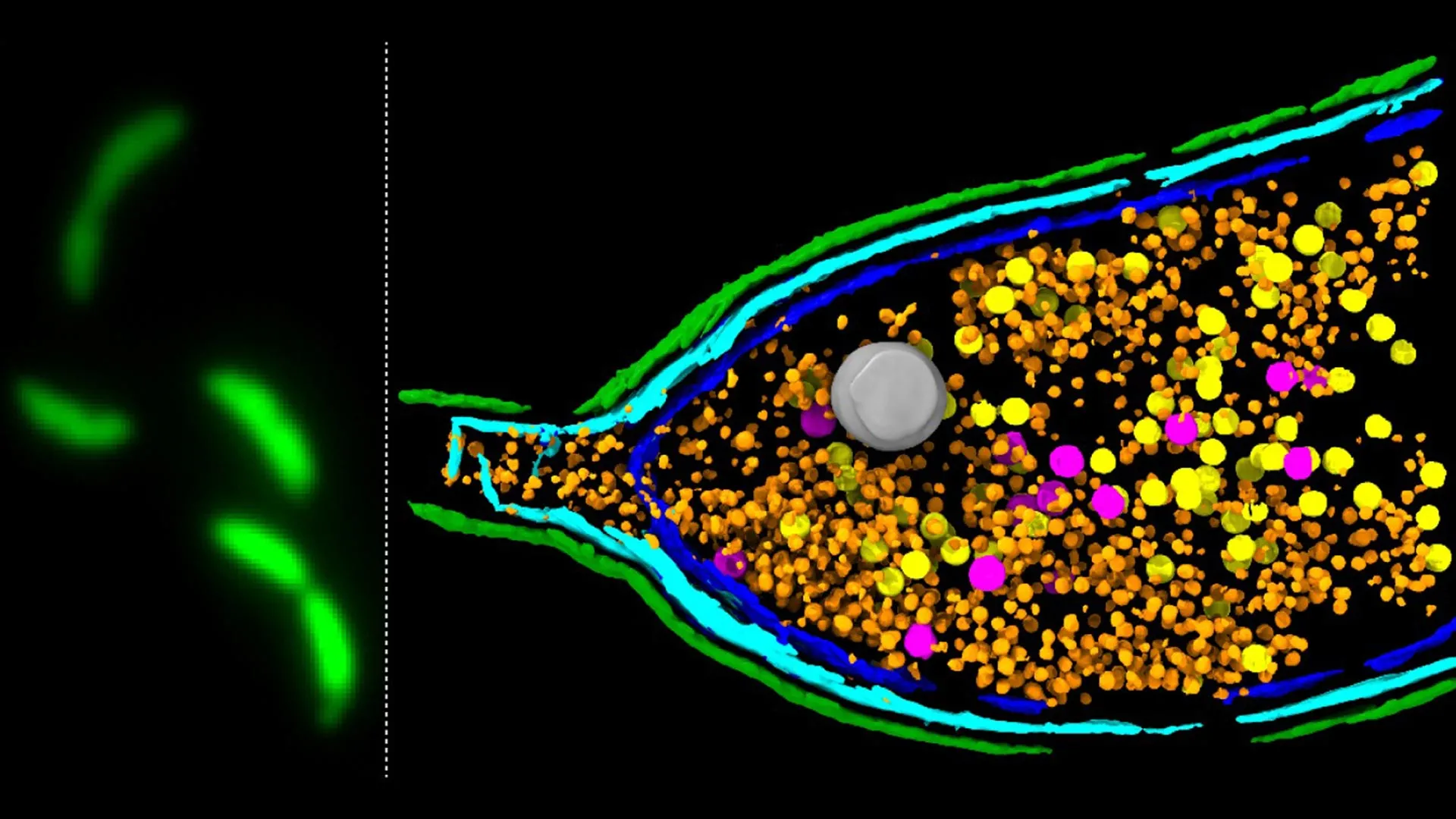
Scientists have unearthed critical new insights into the intricate mechanisms by which bacteria exchange genetic material, a process that significantly contributes to the escalating global health crisis of antimicrobial resistance (AMR). Researchers at the prestigious John Innes Centre have illuminated the previously opaque function of unusual particles known as gene transfer agents (GTAs), revealing how these molecular couriers facilitate the rapid dissemination of traits, including those conferring resistance to life-saving antibiotics.
Unlocking the Secrets of Bacterial Gene Sharing
The groundbreaking research, published in the esteemed journal Nature Microbiology, delves into the fundamental biology of horizontal gene transfer (HGT), a phenomenon that allows bacteria to acquire new genetic information from sources other than their parent cells. This process is a driving force behind bacterial adaptation and evolution, enabling them to overcome environmental challenges and develop resistance to therapeutic interventions.
GTAs, the focus of this investigation, are fascinating entities. They bear a striking resemblance to bacteriophages, viruses that infect bacterial cells. However, the scientific consensus now posits that GTAs are not parasitic invaders but rather ancient viral elements that bacteria have co-opted and domesticated for their own purposes. These self-contained particles, derived from what were once external viral threats, have been repurposed into sophisticated delivery systems for genetic material.
The GTA Mechanism: A Molecular Courier Service
At their core, GTAs function as miniature delivery vehicles. They are capable of encapsulating fragments of the host bacterium’s own DNA and then transporting these genetic payloads to neighboring bacterial cells. This remarkable capacity for gene sharing allows bacterial populations to swiftly acquire and propagate beneficial characteristics, such as the ability to survive exposure to antibiotics.
A pivotal, yet until now poorly understood, stage in the GTA lifecycle is host cell lysis – the programmed rupture of the bacterial cell membrane. This explosive event is essential for the release of the assembled GTA particles, enabling them to embark on their gene-sharing mission. The precise molecular machinery orchestrating this critical lysis event has remained a significant enigma in microbiology.
Pinpointing the Lysis Control Hub: The LypABC System
To unravel this mystery, the research team employed a sophisticated deep sequencing-based screening method. This technique allowed them to systematically identify and analyze the genes responsible for GTA activity within the well-studied model bacterium Caulobacter crescentus. The meticulous analysis led to the identification of a key three-gene system, designated LypABC.
This LypABC system encodes bacterial proteins that, when activated, trigger the lysis of the host cell. The researchers observed that when the genes encoding LypABC were experimentally removed from the bacterial genome, the cells were incapable of rupturing and releasing GTA particles. Conversely, when the LypABC system was deliberately overactivated, a significant proportion of the bacterial population underwent lysis, demonstrating its potent role in controlling cell breakdown. These findings firmly establish LypABC as a central regulatory hub that governs the release of GTAs.
A Surprising Repurposing: From Immune Defense to Gene Transfer
Perhaps one of the most striking revelations from this research is the unexpected functional similarity between the LypABC system and bacterial anti-phage immune mechanisms. Many of the protein components within LypABC are typically associated with cellular defense systems designed to protect bacteria from viral infection. This suggests a remarkable evolutionary adaptation where an existing defense system has been ingeniously repurposed by bacteria to facilitate gene transfer.
This discovery underscores the extraordinary flexibility and adaptability of bacterial biology. It highlights how organisms can retool and reassign biological machinery for novel purposes, blurring the lines between defense and intercellular communication. The collaborative nature of this research, involving the University of York and the Rowland Institute at Harvard, emphasizes the power of interdisciplinary scientific inquiry in tackling complex biological questions.
The Imperative of Tight Regulation
The study also uncovered the existence of a specific regulatory protein that plays a crucial role in maintaining stringent control over GTA activity. This tight regulation is not merely an academic curiosity; it is vital for bacterial survival. The researchers found that improper or excessive activation of the LypABC system can be highly toxic to bacterial cells, leading to widespread cell death. This regulatory oversight ensures that gene transfer occurs in a controlled and beneficial manner, preventing self-inflicted damage to the bacterial community.
Implications for the Global Fight Against Antimicrobial Resistance
The implications of these findings for understanding and combating antimicrobial resistance are profound. The rapid spread of AMR is a critical threat to global public health, jeopardizing our ability to treat common infections and perform routine medical procedures. Bacteria can acquire resistance genes through various mechanisms, with HGT being a particularly efficient route. By shedding light on the precise mechanisms of GTA-mediated gene transfer, this research provides invaluable knowledge for developing novel strategies to disrupt the spread of resistance.
Dr. Emma Banks, the first author of the study and a Royal Commission for the Exhibition of 1851 Research Fellow, commented on the significance of the findings: "What’s particularly interesting is that LypABC looks like an immune system, yet bacteria are using it to release GTA particles. It suggests that immune systems can be repurposed to help bacteria share DNA with each other — a process that can contribute to the spread of antibiotic resistance."
The discovery that bacteria can hijack defense mechanisms for gene dissemination highlights the sophisticated evolutionary strategies employed by these microorganisms. Understanding these processes is paramount for predicting and mitigating the future evolution of resistance.
Future Directions and Ongoing Research
The research team is now focused on delving deeper into the intricacies of the LypABC system. Key areas of investigation include understanding the precise triggers that activate the system and elucidating the detailed molecular pathways through which it orchestrates the rupture of bacterial cells and the subsequent release of GTA particles. This further research could unlock new targets for antimicrobial interventions aimed at inhibiting gene transfer.
A Broader Context: The Evolution of Bacterial Warfare and Cooperation
The evolutionary journey of bacteria has been characterized by a dynamic interplay of conflict and cooperation. For millennia, bacteria have developed intricate defense systems to protect themselves from invaders, including viruses. The discovery of the LypABC system repurposed from an anti-phage defense mechanism exemplifies how these defense strategies can be co-opted and transformed into tools for intercellular communication and genetic exchange. This highlights a fascinating evolutionary paradox: what was once a weapon of defense has become a vehicle for cooperation, albeit cooperation that can unfortunately facilitate the spread of resistance to human medicines.
The historical timeline of our understanding of HGT has progressed from initial observations of transformation and transduction to the more recent appreciation of the widespread role of mobile genetic elements like plasmids and transposons. The identification of GTAs as distinct entities adds another crucial layer to this complex picture. While the exact evolutionary origins of GTAs are still being investigated, their domesticated nature suggests a long history of co-evolution between viruses and bacteria, leading to the current state where these particles serve the interests of their bacterial hosts.
Supporting Data and Scientific Significance
The scientific rigor of the Nature Microbiology publication is supported by robust experimental data. The study utilized quantitative PCR and Western blot analyses to confirm the expression levels of LypABC genes and proteins under different conditions. Furthermore, electron microscopy provided visual evidence of GTA particle formation and release. The use of genetic knockouts and overexpression strains, coupled with advanced sequencing technologies, provided statistically significant and reproducible results, lending strong credence to the conclusions drawn by the researchers.
The significance of this work extends beyond fundamental microbiology. The World Health Organization (WHO) has identified AMR as one of the top 10 global public health threats facing humanity. Projections indicate that by 2050, AMR could cause up to 10 million deaths per year if current trends continue unchecked. Therefore, any research that illuminates the mechanisms of AMR spread holds immense potential for informing public health strategies and the development of new therapeutic approaches.
Expert Reactions and Broader Impact
While specific immediate reactions from external bodies were not detailed in the provided text, the publication of such findings in a high-impact journal like Nature Microbiology invariably sparks interest and discussion within the scientific community. It is highly probable that infectious disease specialists, evolutionary biologists, and pharmaceutical researchers are keenly examining these results for their implications in developing novel anti-AMR strategies.
The broader impact of this research lies in its potential to shift paradigms in how we approach the fight against AMR. Instead of solely focusing on developing new antibiotics, this work suggests that interventions targeting the mechanisms of gene transfer itself could be a viable and potent strategy. Disrupting the ability of bacteria to share resistance genes could significantly slow down the evolution and spread of untreatable infections.
The study’s findings contribute to a growing body of evidence that emphasizes the complex and often counterintuitive ways in which bacteria interact and evolve. It underscores the need for continued investment in basic scientific research to understand the fundamental biological processes that underpin major global health challenges. As the researchers move forward to unravel the activation triggers and control mechanisms of LypABC, the scientific community eagerly anticipates further insights that could pave the way for innovative solutions to one of the 21st century’s most pressing health crises.