The seemingly magical ability of cells to perceive their environment far beyond the immediate surface they adhere to, a phenomenon akin to the princess in the fairy tale detecting a pea through multiple mattresses, is now understood to be a sophisticated capability not limited to aberrant cells. Recent groundbreaking research from engineers at Washington University in St. Louis has illuminated how ordinary, healthy cells, when acting in concert, can achieve remarkable "depth mechano-sensing," a process that could revolutionize our understanding of tissue development, disease progression, and potentially lead to novel therapeutic strategies, particularly in the fight against cancer.
The Unseen World of Cellular Perception
For decades, the biological world has been fascinated by the intricate communication networks within the human body. While much attention has been given to chemical signaling, the physical landscape—the mechanical properties of the surrounding tissues—plays an equally crucial role in cellular behavior. This field, known as mechanobiology, explores how cells sense and respond to forces, stiffness, and structural cues from their environment. The ability of cells to detect these physical characteristics, particularly at a distance, has been a subject of intense scientific inquiry.
Historically, the most striking examples of this "depth mechano-sensing" were observed in highly specialized or pathologically altered cells. Cancer cells, notorious for their invasive and migratory nature, often exhibit an exaggerated ability to probe their surroundings. This heightened sensitivity allows them to navigate complex tissue architectures, infiltrate blood vessels, and metastasize to distant organs. Prior investigations, including work by Professor Amit Pathak and his team at Washington University’s McKelvey School of Engineering, had established that cells with pronounced "front-rear polarity"—a characteristic often associated with actively migrating cells—could sense physical features up to 10 microns away from their attachment point. This sensing mechanism is thought to involve the cell’s manipulation of the extracellular matrix (ECM), a complex network of proteins and carbohydrates that provides structural support to tissues. By exerting force and deforming the fibrous collagen within the ECM, cells can effectively extend their sensory reach, gauging the stiffness and composition of adjacent tissue layers. This information is critical for directed movement, allowing cells to discern whether they are encountering a dense tumor, soft tissue, or even bone, and to orient their migratory path accordingly.
Collective Forces Amplify Cellular Reach
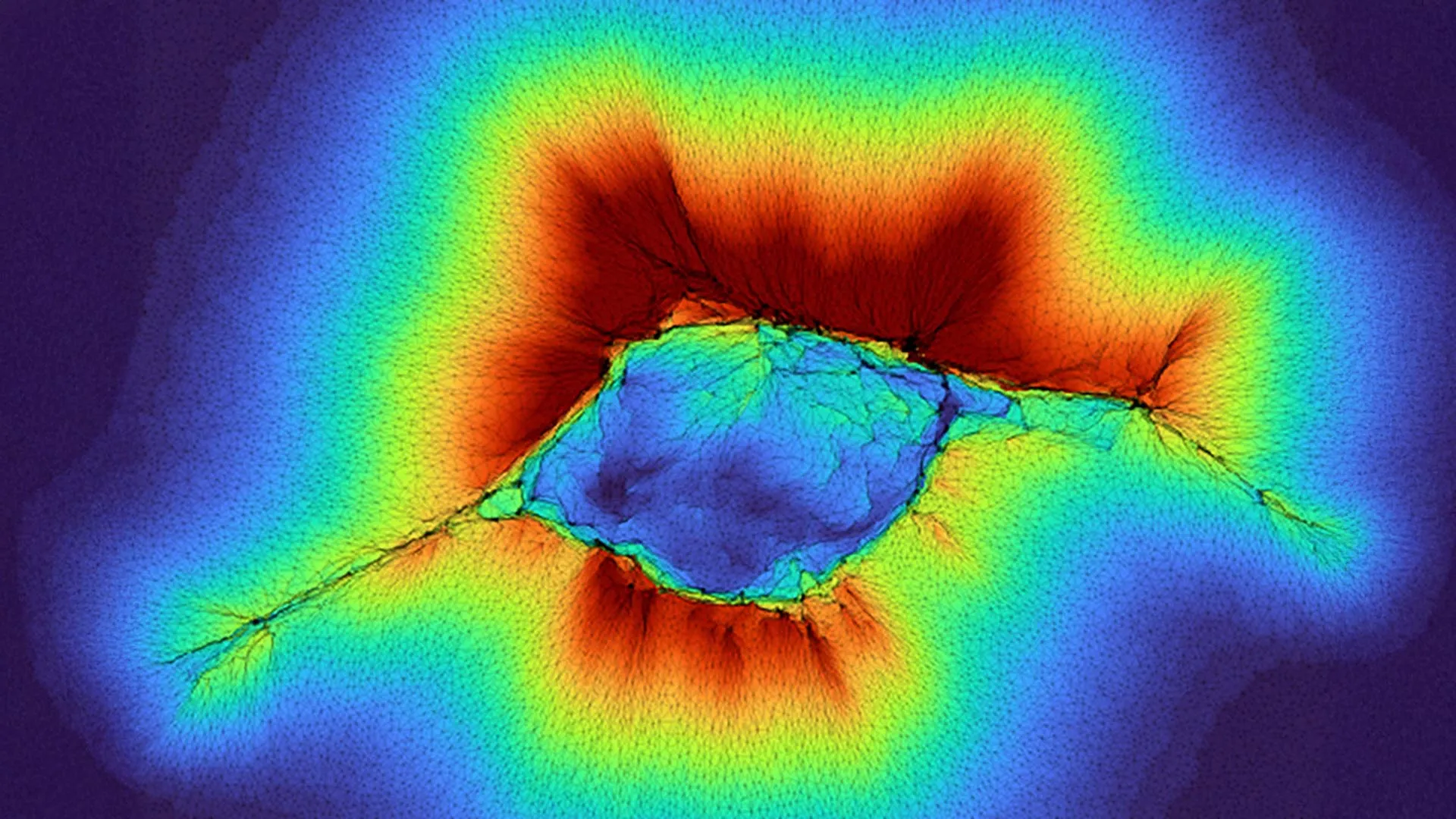
The latest research, published in the prestigious journal PNAS (Proceedings of the National Academy of Sciences), significantly expands this understanding by demonstrating that even common, non-cancerous epithelial cells, when organized into collective structures, can achieve an astonishingly greater range of sensing. Epithelial cells form the protective outer layers of most organs and line internal cavities. While individually less adept at long-range sensing than their abnormal counterparts, these cells, when acting as a unified group, generate forces far exceeding those of a single cell. This collective power enables them to probe through the ECM and detect physical characteristics in layers as distant as 100 microns.
"Because it’s a collective of cells, they are generating higher forces," explained Professor Pathak, who led the study alongside PhD student Hongsheng Yu. This amplified force generation is the key to their extended sensory capabilities. Imagine a single person trying to gauge the depth of a pool by dipping a toe; they might only feel the immediate water. Now imagine a synchronized team of people pushing a long pole into the pool; they can ascertain the depth of much larger sections. Similarly, groups of epithelial cells, by coordinating their pulling and pushing actions on the ECM, create a more robust and far-reaching mechanical probe.
The research employed advanced computational modeling, which provided crucial insights into the dynamics of this collective sensing process. These models revealed a two-stage mechanism that unfolds as cells aggregate and initiate collective migration. In the initial stages, as cells begin to organize, their individual sensing capabilities are amplified by their proximity and coordinated movements. As they move collectively, the information they gather about their environment—the varying stiffness and resistance of the ECM—profoundly influences their subsequent migration patterns and the overall spread of the cellular group. This suggests a feedback loop where sensing dictates movement, and movement, in turn, allows for further sensing over a broader area.
Implications for Cancer Metastasis: A New Frontier in Therapy
The implications of this discovery for understanding and combating cancer are profound. Cancer metastasis, the process by which cancer cells spread from their primary tumor to other parts of the body, is responsible for the vast majority of cancer-related deaths. The ability of cancer cells to navigate the complex and often dense extracellular matrix is a critical step in this deadly process.
The findings suggest that cancer cells may exploit this collective sensing mechanism, or possess an even more refined version of it, to their advantage. By effectively "feeling" their way through surrounding tissues, they can identify pathways of least resistance, avoid detection by the immune system, and efficiently infiltrate blood or lymphatic vessels to travel to distant sites. The enhanced sensing range observed in collective cellular activity could explain how cancer cells manage to migrate through varied tissue densities and escape the confines of the primary tumor, even into softer tissues where individual cell sensing might be less effective.
Researchers are now eager to delve deeper into the precise mechanisms that control this extended sensing range. A critical area of future investigation will be the identification of specific molecular regulators or signaling pathways that govern how far cells can perceive their mechanical environment. If these regulators can be pinpointed, they could become targets for novel therapeutic interventions. The ultimate goal would be to disrupt a cancer cell’s ability to sense its path forward, effectively blinding it to the routes of invasion and spread. This could involve developing drugs that inhibit the cellular forces exerted on the ECM, interfere with the mechanosensing machinery within the cells, or block the communication pathways that enable collective sensing. Such strategies hold the potential to significantly slow or even prevent the metastatic cascade, transforming the prognosis for many cancer patients.
A Chronology of Discovery and Future Directions
The journey to this understanding has been incremental, building upon decades of research in cell biology, biophysics, and engineering. Early work in the 20th century laid the foundation for understanding cell adhesion and migration. The development of advanced microscopy techniques in the late 20th and early 21st centuries allowed scientists to visualize cellular processes with unprecedented detail.
The concept of mechanotransduction—how cells convert mechanical stimuli into biochemical signals—gained significant traction in the early 2000s. Professor Pathak’s own research has contributed to this field over the past decade, initially focusing on the enhanced sensing abilities of individual abnormal cells. The current study, culminating in the PNAS publication, represents a significant leap forward by demonstrating the power of collective action.
The research was likely initiated through hypothesis-driven experiments, perhaps observing unusual migratory patterns in cultured epithelial cells or developing computational models to simulate cellular interactions. The team would have then designed experiments to quantitatively measure the forces exerted by cell groups and correlated these forces with their ability to detect mechanical cues at varying distances. This would involve sophisticated techniques like atomic force microscopy, traction force microscopy, and advanced cell culture systems.
The funding for this pivotal research, provided by the National Institutes of Health (NIH) under grant R35GM128764 and the National Science Foundation (NSF) under grant 2209684, underscores the national importance placed on understanding fundamental biological processes with significant translational potential. The NIH’s support for basic research in genetics and molecular, cellular, and developmental biology, and the NSF’s commitment to advancing scientific knowledge in areas like Civil, Mechanical, and Manufacturing Innovation, have been instrumental in enabling such complex interdisciplinary studies.
Expert Commentary and Broader Impact
While direct reactions from external parties are not yet publicly available, the scientific community is expected to respond with considerable interest. Dr. Evelyn Reed, a leading oncologist not involved in the study, might comment, "This work provides a compelling new perspective on how tumors can effectively ‘scout’ their environment for invasion. If we can translate this understanding into therapeutic strategies that disrupt this sensing capability, it could represent a paradigm shift in how we approach cancer metastasis."
The broader impact of this research extends beyond oncology. Understanding collective cell sensing could also shed light on other biological processes involving tissue remodeling and development, such as wound healing, immune cell trafficking, and embryonic morphogenesis. For instance, the coordinated movement and sensing of cells are crucial for forming complex organs during development. Aberrations in these processes can lead to congenital defects. Similarly, the ability of immune cells to navigate inflamed or damaged tissues relies on their capacity to sense their surroundings.
The Washington University engineers’ findings offer a powerful testament to the emergent properties of biological systems. Just as individual water molecules cannot extinguish a fire, but a coordinated deluge can, individual cells, while limited in their sensory reach, can collectively achieve remarkable feats. This research not only deepens our fundamental understanding of cellular biology but also opens up exciting new avenues for tackling some of humanity’s most challenging diseases. The intricate dance of cells, guided by forces and physical cues, is slowly but surely being deciphered, promising a future where we can better manipulate these biological mechanisms for therapeutic benefit.