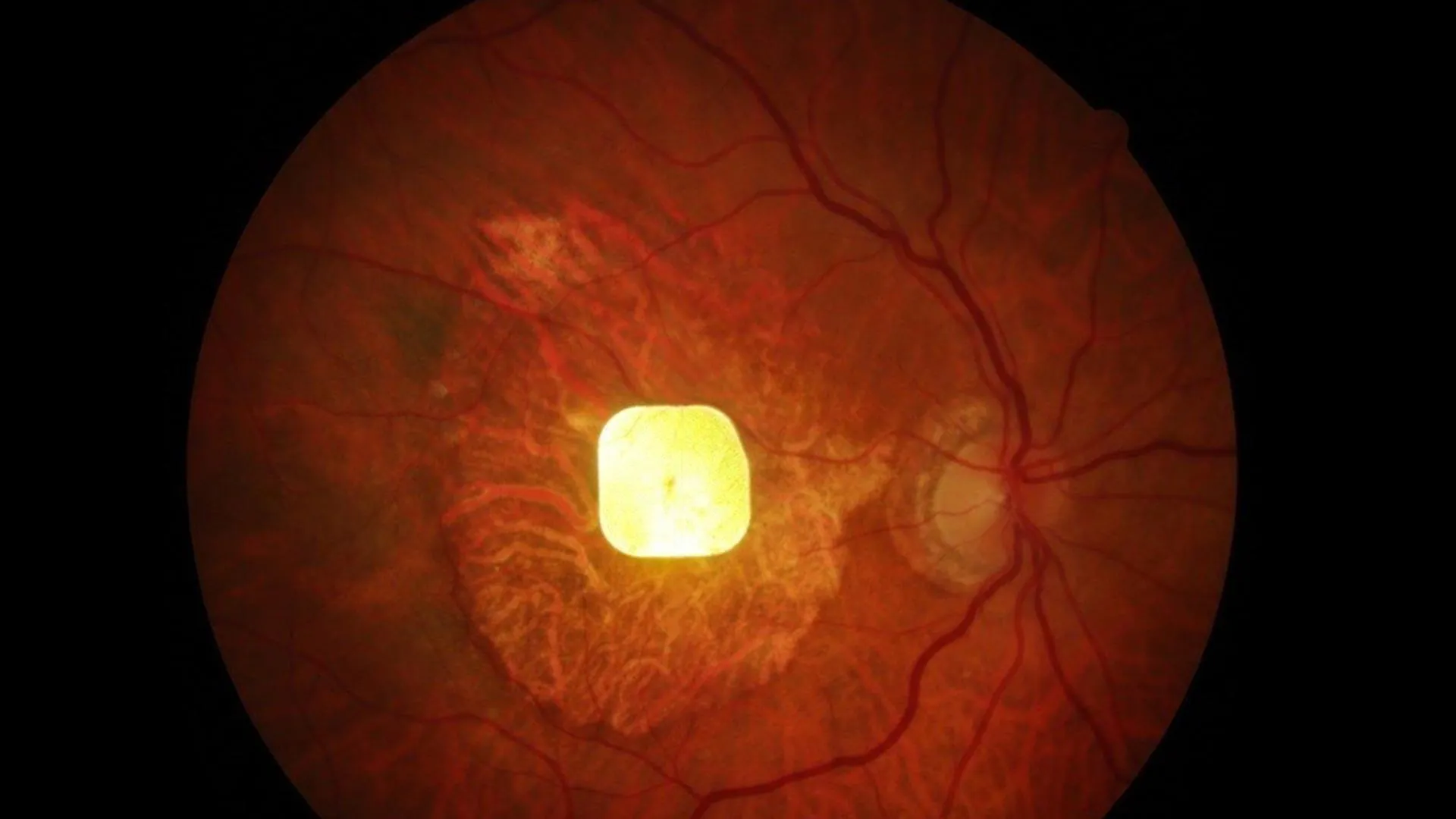
A groundbreaking wireless retinal implant, no larger than a speck of dust, has demonstrated significant success in restoring central vision for individuals suffering from advanced age-related macular degeneration (AMD), specifically the atrophic form known as geographic atrophy (GA). The pivotal findings, published in the prestigious New England Journal of Medicine, offer a beacon of hope for the millions worldwide affected by this leading cause of irreversible blindness in older adults, a condition impacting over 5 million people globally.
A Decade-Long Journey Towards Vision Restoration
The development and rigorous testing of this innovative device, known as the PRIMA system, represent the culmination of over 15 years of dedicated research. Spearheading this international, multi-center clinical trial were three distinguished leaders in ophthalmology: José-Alain Sahel, M.D., director of the UPMC Vision Institute; Daniel Palanker, Ph.D., professor of ophthalmology at Stanford University; and Frank Holz, M.D., professor of ophthalmology at the University of Bonn, Germany. Their collaborative efforts have navigated the complex scientific and clinical landscape to bring this life-changing technology to fruition.
The journey began with the fundamental understanding of how AMD devastates vision. In healthy eyes, specialized light-sensing cells called photoreceptors in the retina capture light and convert it into electrical signals. These signals are then relayed through a complex neural network to the brain, where they are interpreted as images. However, in advanced AMD, particularly GA, these vital photoreceptors in the central part of the retina are progressively and irreversibly damaged. This cellular degeneration leads to blurred or lost central vision, significantly impairing daily activities such as reading, recognizing faces, and navigating familiar environments.
Remarkable Vision Gains Demonstrated in Clinical Trial
The PRIMAvera trial, an international study conducted across 17 medical centers in five European countries – France, Germany, Italy, the Netherlands, and the United Kingdom – enrolled 38 participants aged 60 and older who had been diagnosed with advanced dry AMD with geographic atrophy. The results from the 32 participants who completed one year of follow-up are nothing short of remarkable. A substantial 81% of these participants, equating to 26 individuals, reported meaningful improvements in their visual acuity.
Further underscoring the functional benefits of the implant, 84% of the participants (27 individuals) confirmed that they were actively using the artificial vision provided by the device in their daily lives. This included tasks as fundamental as reading numbers and words, activities that had previously been severely compromised or rendered impossible.
On average, patients who received the implant experienced a significant gain of 25 letters on a standard eye chart. This improvement is equivalent to approximately five lines of text, representing a substantial leap in their ability to perceive visual detail. The threshold for clinically significant improvement is often considered to be a gain of 10 letters; in this trial, an impressive 81% of participants surpassed this benchmark.
Dr. Sahel, the senior author of the study and chair of the Department of Ophthalmology at the University of Pittsburgh School of Medicine, expressed profound optimism regarding these outcomes. "It’s the first time that any attempt at vision restoration has achieved such results in a large number of patients," he stated. "More than 80% of the patients were able to read letters and words, and some of them are reading pages in a book. This is really something we couldn’t have dreamt of when we started on this journey, together with Daniel Palanker, 15 years ago."
The Ingenious Technology Behind the PRIMA System
The PRIMA system’s ingenious design addresses the core problem of photoreceptor loss by offering a sophisticated, yet miniaturized, solution. Developed by Professor Palanker, the wireless implant is a tiny device measuring just 2×2 mm. Its primary function is to bypass the damaged photoreceptors and directly stimulate the surviving retinal cells.
The system comprises two main components: the retinal implant itself and a pair of specialized glasses. The glasses are equipped with a miniature camera that captures visual information. This captured data is then transmitted wirelessly to the implant through invisible near-infrared light. Upon receiving this light signal, the implant converts it into electrical pulses. These electrical pulses then stimulate the remaining healthy retinal neurons, effectively mimicking the natural process of visual signal generation.
Crucially, the PRIMA system is designed to be user-friendly and adaptable. Individuals can adjust settings such as zoom and contrast on their glasses to optimize their visual experience and enhance clarity. This personalized control allows users to fine-tune their vision according to their specific needs and environmental conditions.
A Glimpse into the Clinical Trial Process
The PRIMAvera trial, initiated in early 2019, followed a meticulously designed protocol. Participants underwent a surgical procedure to implant the device onto the retina. Following implantation, they engaged in a period of rehabilitation and training to adapt to the new visual input and optimize the use of the system. The one-year follow-up period was critical for assessing the long-term efficacy and safety of the implant.
During the follow-up period, all participants were regularly monitored for any adverse events. The study reported that any side effects directly related to the surgical procedure had resolved by the end of the 12-month period, indicating a favorable safety profile.
Broader Implications and Future Directions
The success of the PRIMA system in the PRIMAvera trial marks a significant milestone in the field of vision restoration. While the implant does not restore vision to perfect 20/20 acuity, its ability to restore functional central vision for a majority of participants is a transformative development.
"While we can’t yet restore full 20/20 vision with the implant alone, at UPMC we are investigating methods that could further improve people’s quality of life and take them above the threshold for legal blindness," Dr. Sahel remarked. This statement highlights the ongoing commitment to advancing the technology and exploring complementary strategies to further enhance visual outcomes.
The implications of these findings extend beyond individual patient benefits. The development of a wireless, implantable device that can effectively bypass damaged retinal cells opens new avenues for treating other forms of retinal degeneration where photoreceptor loss is a primary issue. It signifies a shift towards more sophisticated bioelectronic interfaces that can integrate with the nervous system to restore lost function.
Regulatory Pathways and Ongoing Research
Buoyed by these positive clinical results, the device manufacturer, Science Corporation (formerly Pixium Vision SA), has taken significant steps towards making the PRIMA system accessible to a wider patient population. Applications have been submitted seeking regulatory approval for clinical use in both Europe and the United States.
The UPMC Vision Institute played a pioneering role in the U.S. context, becoming the first center to implant the PRIMA device in 2020. This initial U.S. study was led by Associate Professor of Ophthalmology Joseph Martel, M.D., further solidifying UPMC’s position at the forefront of ophthalmic innovation.
The collaborative nature of this research is evident in the extensive list of contributing institutions. Beyond the lead centers, the study involved researchers from The University of Bonn; The Adolphe de Rothschild Foundation Hospital and The 15-20 National Eye Hospital in Paris; Moorfields Eye Hospital in London; and the University of Rome Tor Vergata, among others. This broad international collaboration underscores the global effort to combat vision loss.
The PRIMAvera trial was supported by funding from Science Corporation, headquartered in Alameda, California. This financial backing has been instrumental in enabling the extensive research, development, and clinical testing required to bring this complex technology to the cusp of widespread availability.
A Future Illuminated by Technological Advancement
For individuals living with the debilitating effects of advanced AMD, the PRIMA retinal implant represents more than just a medical device; it signifies a renewed possibility of independence and an improved quality of life. The ability to read, recognize loved ones, and engage more fully in daily activities can have profound psychological and social benefits. As regulatory approvals are pursued and manufacturing scales up, the prospect of this technology becoming widely available offers a powerful testament to the relentless pursuit of scientific advancement in restoring sight and combating blindness. The journey from fundamental research to a functional implant has been long and arduous, but the success of the PRIMA system in the PRIMAvera trial has illuminated a path towards a brighter future for millions.